Substances that donate their hydrogen ion and accepts an electron from other substances are called acids. Acids have a pH less than 7.0. On the contrary, substances which accept the proton or hydrogen ion and donate electron are called bases. They have a pH more than 7.0. Acids are sour and bases are bitter by taste.
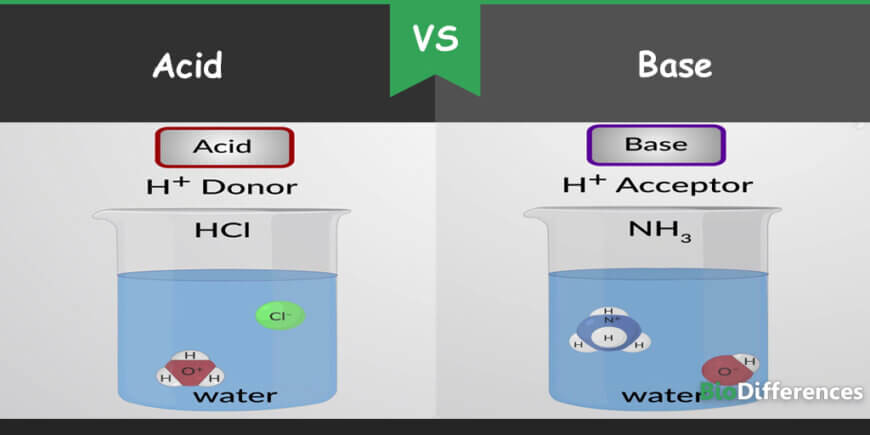
Acid and base are present everywhere, from the human body (nucleic acid and gastric acid) to soaps used for the shower to vinegar present in the kitchen. Acid and base occupy a significant part not only in chemistry but also in other fields of science. Arrhenius theory, Lewis theory, and Bronsted-Lowry theory differentiate both from each other. However, both react with each other to form salt and water.
Contents
Comparison Chart
| Basis for Comparison | Acid | Base |
| Arrhenius Concept | Acid is a chemical when dissolved in water increases the hydrogen ions (H+) concentration in solution. | The base is a chemical when dissolved in water increases the hydroxyl ion (OH–) ions in solution. |
| Lowry-Bronsted Concept | Acids are proton donors | Bases are proton acceptors |
| Lewis Concept | Substances which accepts electron (electrophile) and have vacant orbitals are termed as Lewis acids | Substances that donate electron (nucleophile) and have lone pair of electrons are termed as Lewis bases |
| Chemical Formula | The chemical formula starts with H except for acetic acid. | The chemical formula ends with OH. |
| pH value | Less than 7 | More than 7 |
| Taste | Sour | Bitter |
| Sensation on touch | Sticky | Slippery |
| Reaction with metals | Acids produce hydrogen gas with metals | Bases react with fats and oils. |
| Litmus test | Acids turn blue litmus paper to red | Bases turn red litmus paper to blue |
| Phenolphthalein indicator | Colorless | Pink color |
| Strength | Depends on the concentration of hydronium ions | Depends on the concentration of hydroxide ions |
| Dissociation with water | Acids liberate hydrogen ions after dissociation with water | Bases liberate hydroxide ions after dissociation with water |
| Examples | Nitric acid, hydrochloric acid, sulfuric acid, carbonic acid | Ammonium hydroxide, sodium hydroxide, calcium hydroxide |
What is Acid?
The word acid has derived from the Latin word “acre” which means “sour.” An acid is a chemical that accepts electron and donates H ions or proton. Most acids containing hydrogen-bonded atoms dissociate to give cation and anion in water. Acidity is measured by the presence of hydrogen ion. A higher concentration of hydrogen ions indicates the highest acidity. But high acidity is represented by the low level of pH on the pH meter scale. Acids show a 1-7 pH on the pH meter scale.
Types
Acids can be divided into two types based on the dissociation in water; strong acids and weak acids. Strong acids dissociate completely in water, for example, nitric acid, hydrochloric acid, etc. Weak acids are the acids that are not dissociated completely in water, for example, hydrofluoric acid, acetic acid, etc.
Theories
According to Arrhenius’s theory, an acid can be defined as the substance when added in water, enhances the concentration of hydrogen ions. Bronsted-Lowry did not disagree with the Arrhenius concept of acid; he just added that acid is said to be as the proton donor. It defined the acids without dissolving in water so it is widely accepted and used. According to Lewis’s theory, the compounds which allow the electron pair to form a covalent bond are called acids. But there are some substances that do not contain hydrogen atom but accept electron pair. So this concept is not readily acceptable.
Uses
Acids play many vital functions in the body. DNA and RNA are biological nucleic acids that contain genetic information and hereditary material for transfer from one generation to another generation. Amino acids are another type of acids that help in protein synthesis. Fatty acids and derivatives are the groups of carboxylic acids that also play a vital role in the human body.
Hydrochloric acid is the part of gastric acid in the stomach and helps indigestion. It hydrolysis proteins and polysaccharides. Acids are also present in ants (formic acid) and used in defense mechanisms. Octopi produce a black acid called melanin. Ascorbic acid is present in citrus fruits and very useful for skin. Similarly, lactic acid, sulfuric acid, vinegar, and citric acid are found naturally and have many uses.
What is Base?
Bases are chemicals having characteristics just opposite to the acids. These chemicals reduce the concentration of hydronium ions in the water while acids increase the concentration. Bases donate electrons and accept H+ ions or protons. Bases show pH range 7-14 in pH meter scale. Some strong acids also show behavior as bases.
Types
There is confusion between base and alkali. Mostly bases do not soluble in water, but if a base is dissolved in water, it is called an alkali. Sodium hydroxide acts as a base as well as an alkali. Because it is soluble in water and neutralizes the acids in reactions with acids. While copper oxide is a base but not an alkali. Bases are of different types; strong base, weak base, neutral base, super base, and solid base. Strong bases are completely dissociated into their ions and remove protons from acids for example sodium hydroxide and potassium hydroxide. Weak bases are completely dissociated in water. Neutral bases form a bond with neural acid. Super bases are another type of bases which are formed by alkali metals with their conjugate acids. Solid bases are active in solid forms and used in anion exchange in resins or for reactions with gaseous acids.
Theories
According to Arrhenius’s theory, the species which produces hydroxide ions in water solution is called the base. For example, sodium hydroxide dissociates in water to give sodium and hydroxide ions. But this theory was limited to the chemicals having hydroxide in their structures. According to Bronsted-Lowry theory, a substance that accepts hydrogen ions or protons is called as a base. Lewis’s theory is another widely accepted theory. According to this, an atom with a lone pair of electrons can be said as Lewis base or nucleophile. With the help of lone pairs, these atoms attack the positive charge of the molecule.
Uses
Bases have wide applications and use. Sodium hydroxide is used in the preparation of paper, fiber, and soap. Calcium hydroxide is used as bleaching powder. Magnesium hydroxide is used as an antacid in medicines for digestive problems. It reduces the acidity produced by gastric acids in the stomach. Sodium carbonate is used in the manufacturing of washing soda and for softening the hard water. Sodium hydroxide is also used in the preparation of baking powder as baking soda and the fire extinguisher. Ammonium hydroxide is also used to remove stains from the cloths.
Key Differences
- Acids have a pH value lower than 7.0, whereas bases have a pH value greater than 7.0.
- In the phenolphthalein indicator, acids remain colorless, whereas bases give pink color.
- Acids change the blue color of the litmus paper to red, whereas bases turn red litmus paper to blue.
- Acids are sour, whereas bases are bitter.
- The strength of acids depends on the amount of hydronium ions, whereas the strength of bases depends on the concentration of hydroxide ions.
- Acids conduct electricity whereas bases conduct electricity on dissociation.
Conclusion
In conclusion, acid and bases are different chemicals in characteristics, and both have wide applications in everyday life.